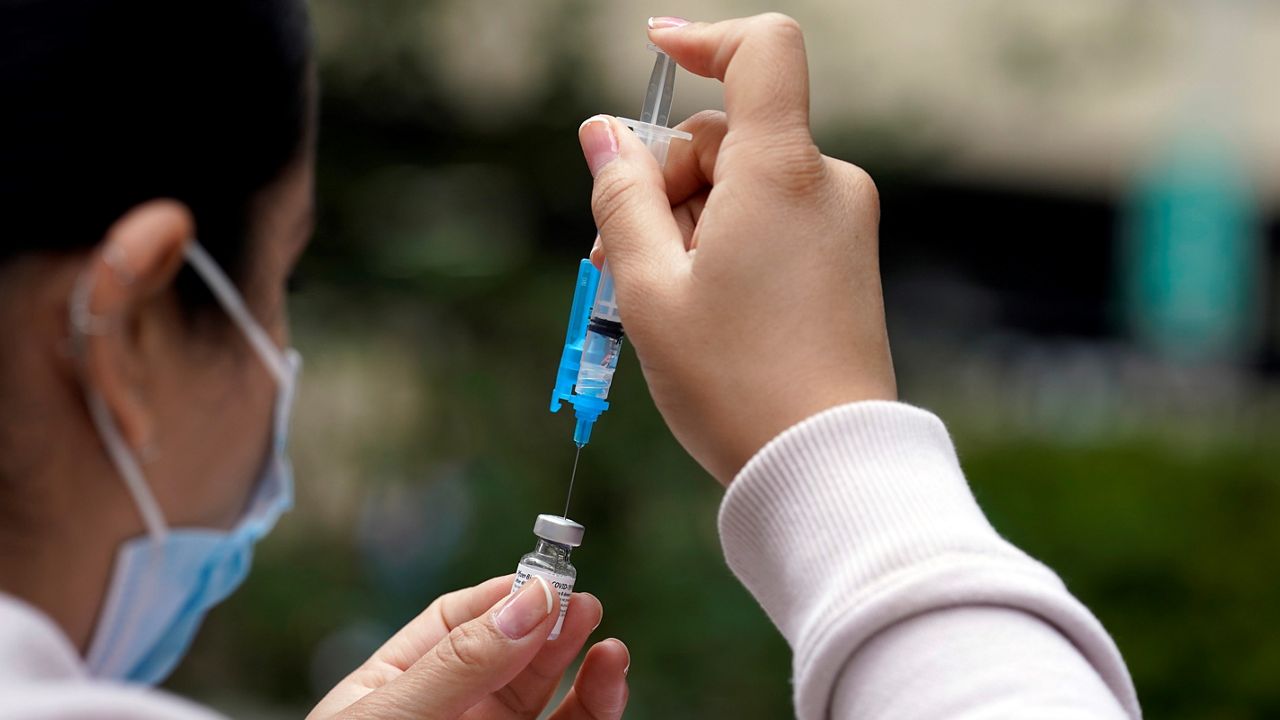
TAMPA — With full FDA approval of Pfizer’s COVID-19 vaccine, mandates have more legal standing whether they come from the government or businesses.
What You Need To Know
- Full approval means more mandates on the way
- Pfizer can now advertise its vaccine
- "Off-label" prescriptions can come from doctors
- RELATED: FDA grants full approval to Pfizer-BioNTech COVID-19 vaccine
On Monday, the Pentagon announced they will require all troops to get vaccinated.
Another consequence of FDA full approval is it gives Pfizer the ability to advertise their product.
Once the pandemic is past us, Pfizer can continue to provide the vaccine which will now be branded as Comirnaty.
Full approval also means doctors could use the vaccine off-label as a booster shot.
USF Professor of Medicine Dr. Michael Teng explained it gives more legal protection to the pharmaceutical company and doctors have more liberty to make vaccinations decisions with their patients.
"So when they print out the label, it's going to say 16 and up, two doses, three weeks apart,” said Dr. Teng. “In this case, off-label using it as a booster, the law gives the doctor more leeway in determining what's right for their particular patient."
For example, Dr. Teng said a doctor can prescribe the booster shot to an elderly patient, even if that patient is not immuno-compromised.
He said they can meet the requirements to get the additional dose to better protect against the delta variant.
The FDA already approved boosters for people with compromised immunity.
Israel has already deployed a booster shot to its elderly population.
As far as what the doctor thinks about Pfizer vaccine's brand name?
Dr. Teng said not to pay much too much attention to that.
"Fact of the matter, nobody uses the proper names for the influenza vaccines either, right?" posited Dr. Teng.
The brand name has been around since the vaccine received its Emergency Use Authorization last December.
Pfizer said in a press release the name, Comirnaty, is a combination of the terms COVID-19, MRNA, community, and immunity.