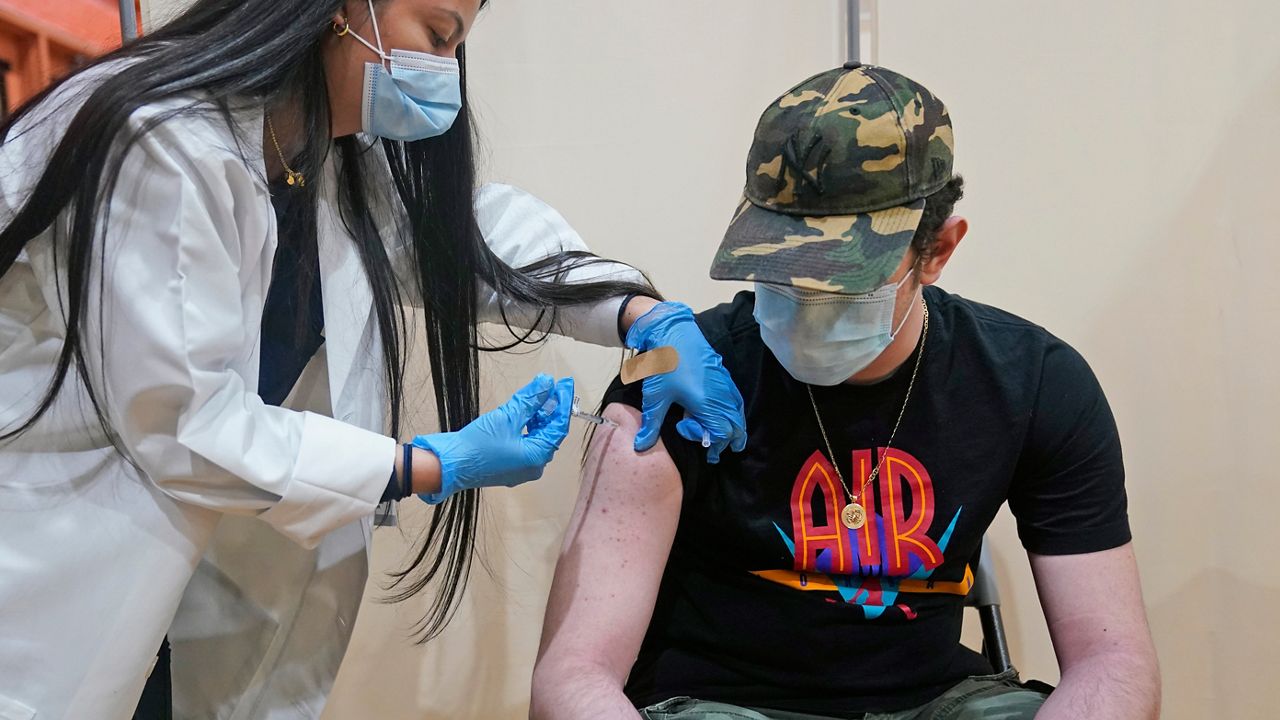
According to multiple reports, the Food and Drug Administration is expected to authorize booster doses of Pfizer-BioNTech’s COVID-19 vaccine for kids 12-15, a welcome sign as pediatric hospitalizations from the coronavirus are on the rise nationwide.
The news was first reported by the New York Times and later confirmed by CNN, both outlets citing individuals familiar with the agency’s plans.
The FDA told Spectrum News that they have no new information to share regarding updated booster guidance.
The reports come as children being hospitalized with COVID-19 are approaching near-record levels.
During the week of Dec. 21-27, on average, 334 children 17 years of age and younger are being hospitalized with COVID-19, according to data from the Centers for Disease Control and Prevention. That’s an increase of 58% from the week prior.
Currently, people as young as 16 are eligible for COVID-19 booster shots. People in the 12-15 age group have been able to get vaccinated with the two-dose since May, meaning millions of adolescents fully vaccinated by July could be eligible for a booster immediately.
The Times reports that the FDA also plans to shift the booster schedule from six months after being fully vaccinated to five months, and authorize a booster for kids 5-11 who are immunocompromised.
An advisory panel to the CDC is expected to meet next week to vote on recommending the changes; if they do so, it’s up to CDC Director Rochelle Walensky to sign off on the recommendation before the changes go into effect.
Dr. Walensky said in an interview with CNN on Wednesday that the agency will "swiftly follow" the guidance of the CDC on boosters for the 12-15 age group.
More than 71% of people 12 and older in the U.S. are fully vaccinated, according to data from the CDC.