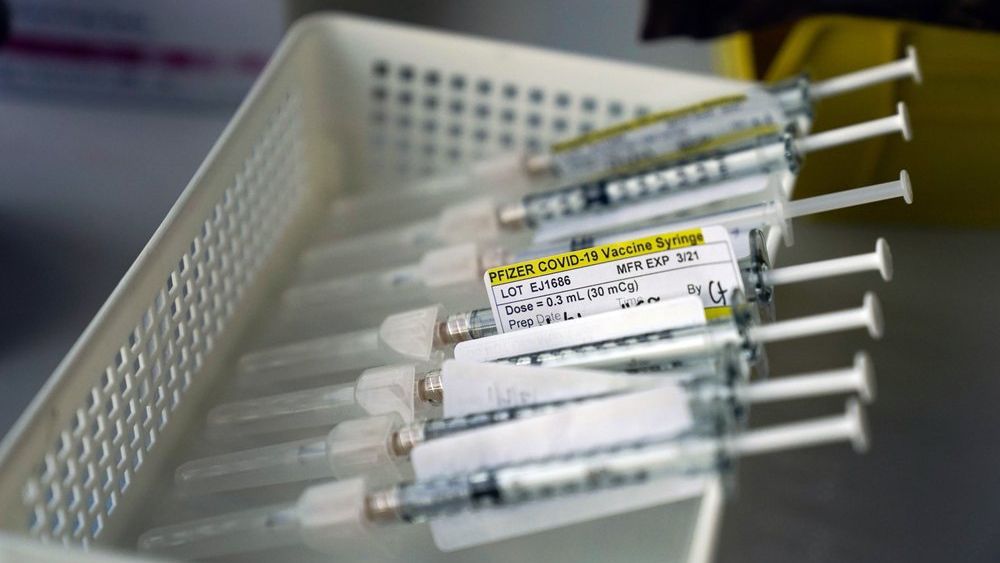
President-elect Joe Biden will release most available COVID-19 vaccine doses to speed delivery to more people, a reversal of the Trump administration policy, his office said Friday.
“The president-elect believes we must accelerate distribution of the vaccine while continuing to ensure the Americans who need it most get it as soon as possible,” spokesman T.J. Ducklo said in a statement. Biden “supports releasing available doses immediately, and believes the government should stop holding back vaccine supply so we can get more shots in Americans’ arms now.”
Under the Trump administration’s approach, the government has been holding back a supply of vaccines to guarantee that people can get a second shot, which provides maximum protection against COVID-19. Both the Pfizer-BioNTech and Moderna vaccines require a second shot about three weeks after the first vaccination. One-shot vaccines are still undergoing testing.
After a glow of hope when the first vaccines were approved last month, the nation’s inoculation campaign has gotten off to a slow start. Of 29.4 million doses distributed, about 5.9 million have been administered, or 27%, according to the Centers for Disease Control and Prevention.
Biden has already indicated his displeasure. In a speech last week, before his election victory was certified by Congress, the president-elect said he intends to speed up vaccinations by having the federal government take a stronger role to make sure that vaccines are not only available, but that shots are going into the arms of more Americans.
“The Trump administration plan to distribute vaccines is falling behind — far behind,” Biden said. “If it continues to move as it is now, it’s going to take years, not months, to vaccinate the American people.”
The American Hospital Association estimates that the nation would need to vaccinate 1.8 million people a day, every day, from Jan. 1 to May 31, to reach the goal of having widespread immunity by the summer. That’s also called “herd immunity” and would involve vaccinating at least 75% of the population.
Without spelling out details, Biden said his administration will put in place a much more aggressive vaccination campaign, with greater federal involvement and leadership, and the goal of administering 100 million shots in the first 100 days.
He said he and Vice-president elect Kamala Harris have been talking with state and local leaders about a coordinated approach that meshes the efforts of governments at all levels. Among the specifics: opening up vaccination centers and sending mobile vaccine units to hard-to-reach communities.
“The top thing is that there’s not a coordinated national plan,” Biden adviser Dr. Rick Bright told The Associated Press.
Ducklo said Biden will share additional details next week on how his administration will engage the pandemic when he takes office on Jan. 20.
Biden’s plan to change the vaccine distribution plan was first reported by CNN.
The news comes amid promising new research from Pfizer that its vaccine is effective against a mutation found in the two more-contagious variants of the coronavirus that have erupted in Britain and South Africa.
“There’s no reason to think the vaccines won’t work just as well on these strains,” said Dr. Frederic Bushman of the University of Pennsylvania, who tracks how the virus mutates.
The mutated version circulating in Britain has also been detected in the U.S. and numerous other countries. That and the variant seen in South Africa are causing global concern because they appear to spread more easily — although how much more isn’t yet known.
The other vaccines now being used in the West — Moderna’s and AstraZeneca’s — are undergoing similar testing.
The new study looked at one modification to the spike protein that both variants share, called N501Y, that is believed responsible for the easier transmission. Pfizer teamed with researchers from the University of Texas Medical Branch in Galveston for laboratory tests to see if that mutation could thwart its vaccine.
They used blood samples from 20 people who received the vaccine, made by Pfizer and its German partner BioNTech, during a large trial of the shots. Antibodies from those recipients fended off the virus in lab dishes, according to the study, posted late Thursday on an online site for researchers.
In a statement released Friday, the FDA said that it is monitoring the potential impact of viral mutations on COVID-19 tests.
“The FDA will continue to monitor SARS-CoV-2 genetic viral variants to ensure authorized tests continue to provide accurate results for patients,” said FDA Commissioner Stephen M. Hahn, M.D in a statement. “While these efforts continue, we are working with authorized test developers and reviewing incoming data to ensure that health care providers and clinical staff can quickly and accurately diagnose patients infected with SARS-CoV-2, including those with emerging genetic variants. At this time, we believe the data suggests that the currently authorized COVID-19 vaccines may still be effective against this strain. The FDA will continue to keep health care providers and the public informed of any new information as it becomes available.”
The FDA said that three authorized testing kits "may be impacted by genetic variants of SARS-CoV-2, but the impact does not appear to be significant."
The Associated Press contributed to this report.